Use Case
Employee Monitoring for SOP Compliance: Verify Digital Process Adherence Without Manual Auditing
Employee monitoring for SOP compliance is the practice of using workforce activity data — application usage logs, sequence records, and timestamped activity trails — to verify that employees are following Standard Operating Procedures in their digital workflows, without requiring supervisors to manually observe each task or rely on self-reported compliance records. Industries from pharmaceutical manufacturing to financial services use this approach to generate objective, auditable evidence of process adherence at scale.
Trusted by 1,000+ companies. Setup in under 2 minutes.
What Is Employee Monitoring for SOP Compliance, and Why Do Regulated Industries Need It?
Standard Operating Procedures define precisely how work should be performed in regulated environments. An SOP specifying that every customer interaction must be logged in the CRM system before the ticket is closed is a process control — a requirement, not a suggestion. But traditional compliance verification relies on supervisors observing individual employees, reviewing sample records, or trusting that employees follow procedures they were trained on. None of these methods scale, and all of them are retrospective: they discover non-compliance after it has occurred.
Employee monitoring for SOP compliance addresses this gap by generating continuous, objective digital evidence of whether procedures are being followed. When the SOP requires opening a specific application, monitoring records whether that application was opened. When the SOP specifies a two-hour documentation window after a patient encounter, monitoring timestamps verify whether the documentation system was accessed within that window. The result is not surveillance — it is an automated compliance audit that runs continuously without consuming supervisor or compliance team time.
The business case is substantial. A single SOP deviation in a pharmaceutical manufacturing context can trigger an FDA Form 483 observation, a recall, or a warning letter. A financial advisor who fails to present required disclosure documents before a trade execution faces FINRA enforcement action that can result in a fine, suspension, or bar. In healthcare, incomplete clinical documentation before patient discharge creates liability under Joint Commission standards and CMS conditions of participation. The cost of consistent SOP non-compliance in these environments vastly exceeds the cost of monitoring infrastructure.
Which Industries Use Employee Monitoring for SOP Compliance Most Effectively?
SOP compliance monitoring applies across any industry where work processes are formalized in written procedures and regulatory or quality frameworks require evidence of adherence. Five industries have developed the most mature use cases.
Pharmaceutical and Biotech (GxP Compliance)
GxP regulations — including Good Manufacturing Practice (GMP), Good Laboratory Practice (GLP), and Good Clinical Practice (GCP) — require documented evidence that defined processes were followed. FDA 21 CFR Part 11 establishes specific requirements for electronic records used as regulatory evidence, including requirements for audit trails, individual user identification, and record integrity. Employee monitoring data meets these requirements when the system is properly validated and access is controlled by unique user credentials.
In a pharmaceutical manufacturing environment, an SOP might require that a quality management system be opened and a batch record reviewed before any production step begins. Monitoring provides an objective log of whether the QMS was accessed, by whom, and when — complementing the paper or electronic batch record with behavioral evidence that the required review occurred.
Financial Services (Trade Compliance and Disclosure Requirements)
Financial services firms operate under extensive procedural requirements from FINRA, the SEC, and state regulators. FINRA Rule 4511 requires broker-dealers to make and preserve records sufficient to demonstrate compliance with applicable regulations. A specific compliance SOP might require that a financial advisor open and present the firm's disclosure document system to the client before executing any trade. Monitoring provides a timestamped record of whether the required disclosure software was accessed during the customer interaction window — objective evidence for compliance examinations.
In wealth management and insurance, suitability documentation SOPs require that specific information be captured in the client record before a product recommendation is made. Monitoring of CRM and documentation system usage during client interaction times verifies that the required documentation step occurred within the required timeframe.
Healthcare (Clinical Documentation Protocols)
Healthcare organizations face SOP compliance requirements under Joint Commission accreditation standards, CMS conditions of participation, and internal quality frameworks. Clinical documentation SOPs define requirements such as: all patient assessments must be documented in the EHR within four hours of the encounter; discharge planning documentation must be completed before the patient's discharge order is entered; medication reconciliation must be completed in the pharmacy system before a patient is transferred between units.
Employee monitoring verifies EHR and clinical system usage at the timestamps required by these SOPs. A compliance team can pull a report showing every patient encounter where the required EHR documentation was not completed within the specified window — generating a compliance exception list without requiring a manual chart review of every record.
Insurance (Claims Processing Procedures)
Insurance companies define detailed claims processing SOPs to ensure consistent decision-making, regulatory compliance, and fraud prevention. A claims adjuster SOP might require that a fraud screening system be queried before any claim above a threshold amount is approved. A personal lines SOP might require that a specific underwriting system be consulted before a rate quote is provided to a customer. Monitoring verifies that the required system was accessed at the required stage of the claims workflow, providing evidence for state regulatory audits and internal quality reviews.
Manufacturing (Quality Management System Adherence)
ISO 9001 and industry-specific quality management frameworks (IATF 16949 for automotive, AS9100 for aerospace) require controlled processes with documented evidence of adherence. In modern manufacturing environments where quality management, production planning, and engineering change order systems are all software-based, monitoring of system usage provides objective evidence that required approvals, reviews, and sign-offs occurred in the correct sequence before production proceeded.
How Does Employee Monitoring Verify SOP Adherence in Practice?
Employee monitoring software verifies SOP adherence through four specific mechanisms. Each mechanism targets a different type of process control requirement found in regulated SOPs.
Mechanism 1: Required Software Usage Verification
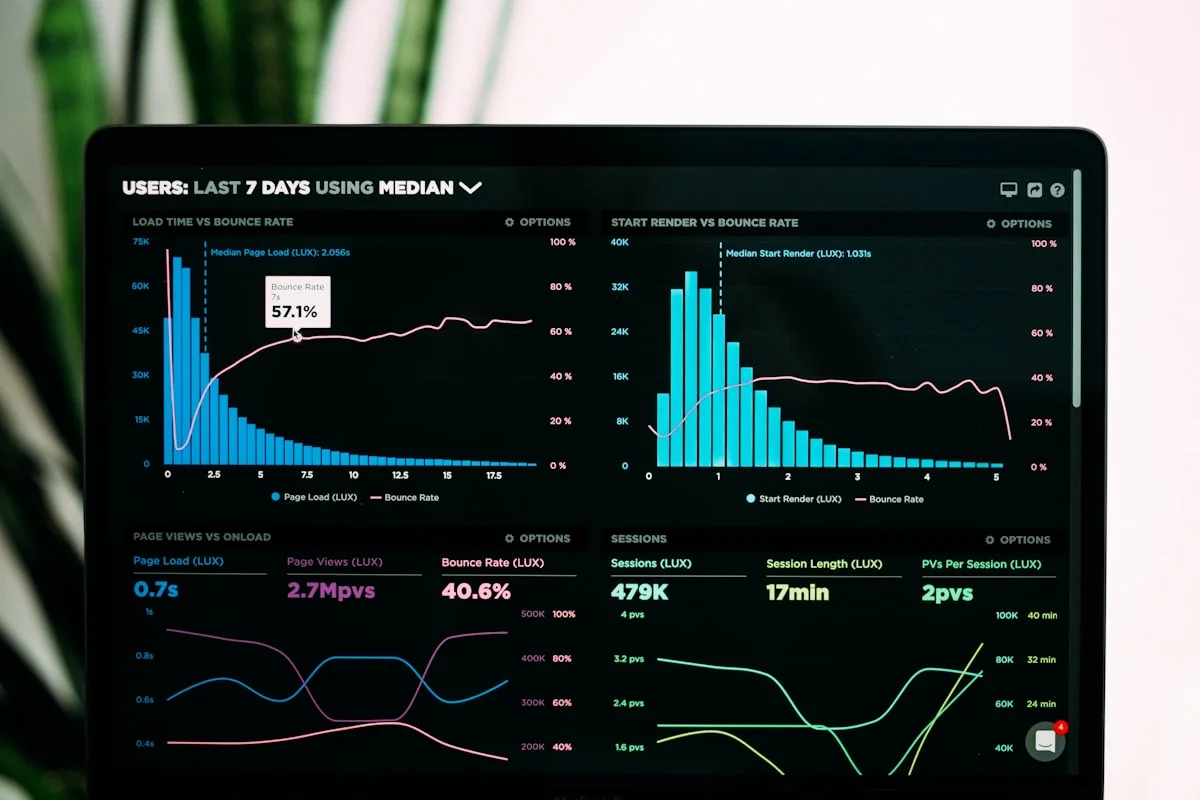
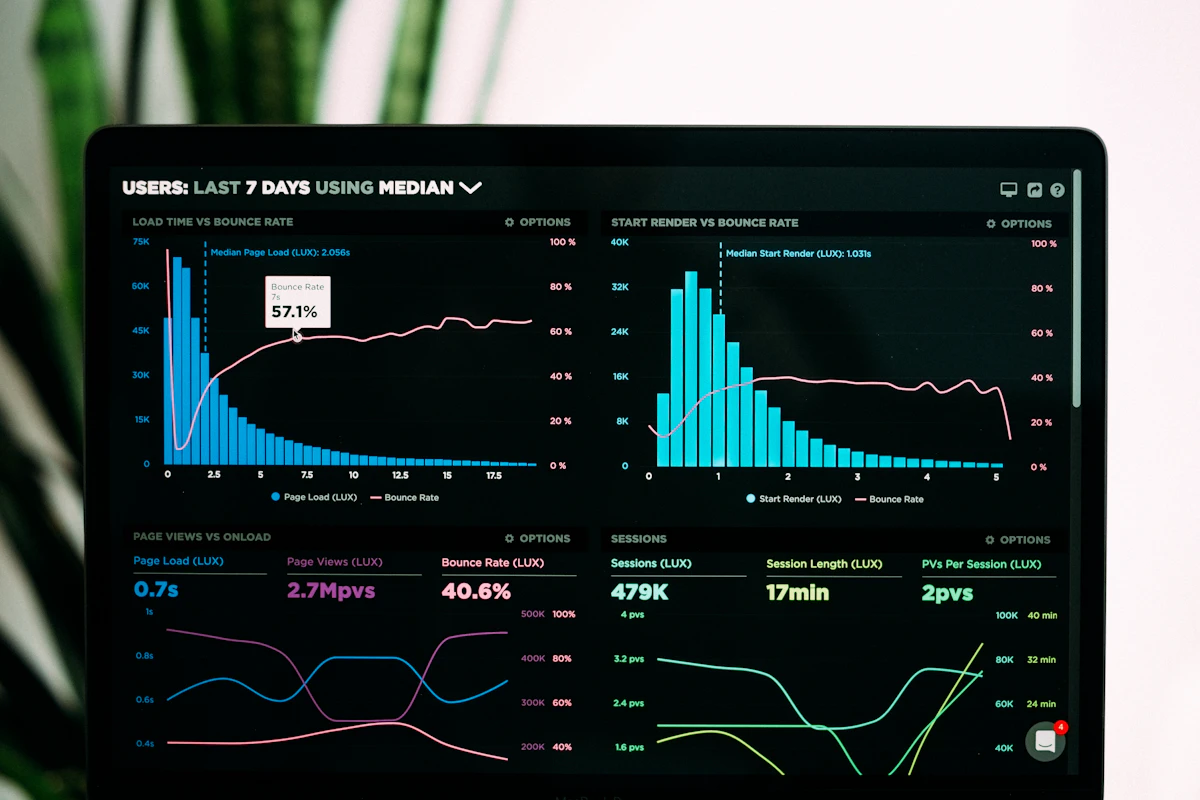
The most common SOP compliance verification use case confirms that required applications were used during specific work activities. The monitoring system tracks which applications are open and active on the employee's workstation throughout the workday. Compliance reporting can show, for any defined time period, every instance where a customer interaction or transaction occurred without the required application being open — identifying potential SOP deviations for compliance investigation.
Practical example: a financial services firm's SOP requires that advisors open the firm's compliance disclosure system before initiating any client call. The compliance team configures a report showing every advisor call event (identified by telephony system activity) during which the disclosure system was not opened. This report runs weekly, identifies exceptions automatically, and generates a list of records requiring follow-up — replacing a manual audit that previously required a compliance analyst to review hundreds of call records individually.
Mechanism 2: Application Sequence Verification
Some SOPs specify not just which applications must be used, but the sequence in which they must be used. Employee monitoring's activity timeline — a chronological, timestamped record of every application session throughout the employee's workday — enables compliance teams to verify that process steps occurred in the required order.
Practical example: a pharmaceutical quality assurance SOP requires that batch documentation be reviewed in the QMS before the production supervisor accesses the execution system to authorize a production run. The monitoring timeline shows whether QMS access preceded execution system access for each production authorization event. Instances where the execution system was accessed before the QMS documentation review are flagged as sequence deviations.
Mechanism 3: Time-Compliance Verification
Many SOPs specify time-based requirements: documentation must occur within a defined window after an event, a review must be completed before a specified deadline, or a system must be updated at defined intervals. Employee monitoring timestamps provide objective evidence of when activities occurred, enabling compliance teams to verify whether the time-based requirements of SOPs were met.
Practical example: a hospital's clinical documentation SOP requires that nursing assessment documentation be completed in the EHR within four hours of patient admission. The monitoring system shows the timestamp when the nurse's workstation first accessed the patient's EHR record, and the timestamp when documentation activity in the EHR was last observed during the four-hour window. Admissions where EHR documentation activity does not appear within the four-hour requirement are identified as potential documentation gaps for clinical quality review.
Mechanism 4: Prohibited Activity Detection
Some SOPs define prohibited activities — behaviors that must not occur during specific work contexts. Monitoring can detect these violations automatically, generating real-time alerts when a prohibited behavior is detected rather than discovering the violation during a retrospective audit weeks later.
Practical example: a customer service center SOP prohibits agents from accessing personal social media or personal email during active customer interactions, as defined by the telephony system showing a customer call in progress. Monitoring of application usage during call sessions detects any personal application access during active calls, generating an immediate alert to the supervisor rather than discovering the policy violation during a random quality audit weeks after the event.
Building an Audit Trail for SOP Compliance Auditors
The value of employee monitoring for SOP compliance extends beyond day-to-day exception detection. When regulators arrive for an inspection or an internal audit team conducts a compliance review, monitoring data provides a historical audit trail that retrospective self-reporting and paper-based records cannot match.
What the Monitoring Audit Trail Contains
eMonitor's activity logs contain, for each employee and each day: every application opened and the duration of each session; the precise timestamp of each application launch and close; the active work sequence showing the order in which applications were accessed; alert history showing any compliance thresholds triggered; and administrator access logs showing who reviewed the monitoring data and when. This data is retained according to your configured retention period (up to the maximum supported by your subscription tier) and is exportable in formats suitable for regulatory review.
Responding to Regulatory Inspection Requests
When a regulatory inspection requires evidence of SOP adherence for a specific period, the compliance team generates a monitoring activity report for the named employees and timeframe. The report shows, chronologically, the applications accessed during each work session — providing objective behavioral evidence to supplement transactional records in regulated systems. In financial services examinations, monitoring logs that corroborate disclosure system usage before trade executions support the firm's compliance attestation. In FDA inspections, electronic records showing QMS access before production authorization events support the batch documentation trail.
Chain of Custody for Monitoring Evidence
For monitoring data to function as credible compliance evidence, the chain of custody must be documented. This means maintaining records of: who has access to the monitoring platform and the specific permissions each role holds; when monitoring data was accessed and for what purpose; any reports generated from the monitoring data and who received them; and any configuration changes that might affect the completeness or accuracy of the monitoring record. eMonitor's immutable admin audit trail satisfies this requirement by logging all access and configuration events with timestamps and user identity.
How to Implement Employee Monitoring for SOP Compliance: A Practical Framework
Successful SOP compliance monitoring programs follow a structured implementation process that connects monitoring configuration directly to the documented requirements of specific SOPs. Deploying monitoring broadly without this connection produces data that is difficult to act on and harder to defend to employees and regulators.
Step 1: Map Your SOPs to Digital Activities
For each SOP you intend to verify through monitoring, document the specific digital activities the SOP requires. List the applications that must be used, the sequence they must be used in (if specified), and any time-based requirements. This map becomes the configuration specification for eMonitor's alert and reporting rules. A financial services SOP that requires disclosure software access before trade execution maps to: monitor for telephony or trading system activity; require disclosure system application session within the same activity block; alert when the required sequence does not occur.
Step 2: Configure Role-Specific Application Classifications
eMonitor's application classification system allows you to define, for each role or team, which applications are required for SOP-covered activities. Configure the required applications for each SOP-covered role so that the compliance reporting distinguishes between general application usage and SOP-required application usage. This prevents false positives in compliance reports and ensures that the monitoring data is interpreted within the correct procedural context.
Step 3: Set Compliance Alert Thresholds
Configure eMonitor's alert system to trigger when the monitoring data indicates a potential SOP deviation. For required software usage SOPs, set alerts for when the required application has not been opened during a defined activity window. For time-compliance SOPs, configure alerts when the monitoring timestamp for required system access falls outside the SOP-specified window. For prohibited activity SOPs, configure real-time alerts when the prohibited application is detected during a monitored work context.
Step 4: Notify Employees and Document the Legal Basis
Employee monitoring for SOP compliance requires transparent advance notification. Update your employee monitoring policy to describe specifically that monitoring data is used to verify SOP adherence, which SOPs are covered, and what the process is when a deviation is detected. For GDPR-regulated organizations, document the legal basis for compliance monitoring — typically Article 6(1)(c) (legal obligation) or Article 6(1)(f) (legitimate interest) depending on whether the SOP is regulatory-driven or internally-mandated. A Data Protection Impact Assessment (DPIA) is recommended for systematic process compliance monitoring that affects a significant number of employees.
Step 5: Establish the Compliance Exception Review Process
Define what happens when monitoring identifies an SOP deviation. Who reviews the exception? What is the investigation process? What documentation is required? What are the potential consequences for confirmed violations versus first-time deviations? The monitoring data identifies potential deviations — the human review process determines whether they represent genuine SOP violations or legitimate exceptions (system downtime, approved procedure workarounds). Document this process in your monitoring policy before deploying the compliance monitoring configuration.
Real-World SOP Compliance Monitoring Scenarios
The following scenarios illustrate how organizations in different industries apply the four monitoring mechanisms described above to specific SOP compliance challenges.
Scenario 1: Financial Advisor Disclosure Compliance
A regional broker-dealer employs 85 financial advisors across three branch offices. The firm's SOP, required under FINRA Rule 2111 (Suitability) and SEC Regulation Best Interest, requires that advisors open and review the firm's disclosure document portal before initiating any client recommendation call. The compliance department previously verified adherence through quarterly random call audits — reviewing 15-20 calls per advisor per quarter, a process requiring 3-4 days of compliance staff time per cycle.
After deploying eMonitor with a compliance monitoring configuration, the firm generates a weekly report showing every advisor call event (identified by telephony system activity in the monitoring log) during which the disclosure portal was not opened within 10 minutes before the call. The report identifies 12 potential exceptions across the firm in the first week — representing a 14.4% exception rate. Investigation reveals seven were legitimate (the advisors had opened the portal on a different device or in a prior session the same day) and five represented genuine SOP deviations requiring documentation and retraining. The monitoring system replaced a quarterly sample audit with a continuous, comprehensive compliance check generating actionable exceptions within 24 hours of the activity.
Scenario 2: Healthcare Clinical Documentation Timeliness
A 250-bed regional hospital faces Joint Commission survey findings related to incomplete nursing assessment documentation. The clinical documentation SOP requires that nursing assessments be entered in the EHR within four hours of patient admission. The compliance team previously identified deviations during chart review — a process that examined 5% of admissions and generated results two weeks after the fact.
eMonitor's activity monitoring tracks EHR system access on nursing workstations. A compliance report generates, for each nursing shift, a list of patient admission events where the assigned nurse's workstation did not log EHR activity within the four-hour documentation window. The weekly report shows 23 out of 340 admissions (6.8%) with potential documentation gaps, significantly higher than the 5% sample audit had indicated. The compliance team reviews the exceptions, confirms 16 are genuine gaps, and refers them to the clinical quality team. The remaining seven have explanations: two were EHR access from a different workstation (not captured in the initial report query), and five were patients transferred from the ED with pre-existing documentation. Adjusting the monitoring query parameters to account for these scenarios improves exception accuracy in subsequent weeks.
Scenario 3: Insurance Claims Fraud Screening
A property and casualty insurance company has an SOP requiring that all claims above $25,000 be screened through the firm's fraud detection platform before a claims adjuster approves payment. The SOP exists to satisfy state regulatory requirements for fraud prevention programs. Prior to monitoring, compliance verification relied on a monthly audit of approved claims above the threshold — checking whether a fraud screen record existed in the platform for each claim.
After deploying eMonitor, the compliance team configures a report showing all claims processing sessions above the threshold (identified by claim amounts in the claims management system) during which the fraud detection platform was not accessed. The daily exception report identifies 8 claims in the first month where fraud screening was not completed before approval — a process gap the monthly audit had not detected because the platform records could be back-dated. The monitoring timestamp evidence shows when each system was actually accessed, providing objective evidence that the fraud screening did not occur before approval regardless of what the platform record showed.
Frequently Asked Questions About Employee Monitoring for SOP Compliance
How does employee monitoring verify SOP compliance?
Employee monitoring software verifies SOP compliance by tracking which applications employees open, in what sequence, and at what times during their work activities. When an SOP requires the use of specific software — a CRM during customer calls, a documentation system before patient discharge, a disclosure platform before trade execution — monitoring data provides an objective record of whether the required software was actually used in the correct timeframe and sequence.
Which industries use employee monitoring for SOP compliance most?
The industries with the most developed SOP compliance monitoring programs are pharmaceutical and biotech (GxP, FDA 21 CFR Part 11), financial services (FINRA, SEC, trade compliance), healthcare (clinical documentation protocols, Joint Commission standards), insurance (claims processing procedures, state regulatory requirements), and manufacturing (quality management systems, ISO 9001 adherence). Any industry with formalized written procedures and regulatory audit requirements benefits from objective digital compliance verification.
Can employee monitoring detect if an employee skipped a required process step?
Employee monitoring detects SOP skip events by identifying gaps in the expected application usage sequence. If an SOP requires opening System A, then System B, then System C before completing a transaction, and monitoring logs show the employee accessed System A and System C without an intervening System B session, that gap is flagged as a potential process deviation. The monitoring audit trail provides timestamped evidence for the compliance investigation that follows.
Is employee monitoring for SOP compliance different from keystroke logging?
Yes. SOP compliance monitoring focuses on application usage patterns, sequence verification, and time-compliance checks — not on what employees type. Monitoring which applications were open and when is significantly less privacy-invasive than logging keystroke content. Most SOP compliance use cases require only application-level activity data, making this monitoring approach appropriate for a wider range of regulated environments where keystroke logging would face privacy objections.
Does monitoring data count as an audit trail for regulatory inspections?
Employee monitoring data constitutes an electronic audit trail when it is timestamped, tamper-resistant, and tied to individual employee identifiers — characteristics that align with FDA 21 CFR Part 11 requirements for electronic records. For financial services, monitoring logs documenting required disclosure software usage before trade execution provide evidence for FINRA Rule 4511 compliance. Consult your compliance counsel to confirm monitoring data meets the specific evidentiary standard for your regulatory framework.
How do you set up SOP compliance monitoring without creating employee privacy concerns?
SOP compliance monitoring is best scoped to the specific activities covered by the SOP — not comprehensive behavioral monitoring. Define exactly which applications are required, during which work activities, and configure alerts only for deviations from those specific requirements. Notify employees in advance about what is monitored and why, linking monitoring to the existing SOP acknowledgment process. Transparency reduces privacy concerns and strengthens the legal basis for monitoring under GDPR's legitimate interest provisions.
What is GxP compliance and how does monitoring support it?
GxP is a collective term for the Good Practice regulations (GMP, GLP, GCP) governing pharmaceutical manufacturing, laboratory operations, and clinical trials. GxP compliance requires documented evidence that defined processes were followed. Employee monitoring supports GxP by providing timestamped records of required software usage, sequence adherence, and work activity during GxP-covered processes — supplementing paper-based records with objective electronic behavioral evidence.
Can employee monitoring prevent SOP violations in real time?
Employee monitoring can generate real-time alerts when an SOP deviation is detected — for example, when a customer service agent closes a call without opening the required documentation system within the SOP-specified timeframe. Real-time alerts allow supervisors to intervene immediately, prompting the employee to complete the required step before the process gap becomes a formal compliance exception in the audit trail.
How does eMonitor support SOP compliance auditing?
eMonitor supports SOP compliance auditing through application usage logs with timestamps, configurable alerts for required application usage thresholds, activity timeline views showing the sequence of applications used during each work session, and exportable audit reports for any specified period. Compliance teams can generate reports showing every interaction handled without required application usage during a specific period — replacing manual chart reviews with automated exception identification.
Do employees need to be informed that monitoring is used for SOP compliance?
Yes. Employees must be informed in advance that monitoring data is used to verify SOP adherence. Notification should appear in the SOP itself, the employee monitoring policy, and the onboarding materials for roles subject to compliance monitoring. Under GDPR, the legal basis for compliance monitoring — typically legitimate interest under Article 6(1)(f) or legal obligation under Article 6(1)(c) — must be documented. Monitoring employees for compliance without notification creates legal exposure in every regulatory jurisdiction.
What is the difference between SOP compliance monitoring and performance monitoring?
SOP compliance monitoring verifies that required process steps were followed — binary adherence to defined procedures. Performance monitoring measures how efficiently and effectively an employee works across all their activities. The two purposes can coexist in the same monitoring platform but require separate policies. Compliance monitoring data should not be repurposed for performance evaluations without explicit policy authorization and employee notification.